On the other hand, the extremely high speed of electrons (unlike that of planets) suggested that relativity be brought into the picture. r We have seen that in Bohr’s model the coulombic force is assumed to be equal and opposite to the centrifugal force equation (1). On the one hand, the positive analogy suggested that electrons (like planets) revolved around the center of mass of the atom (solar system). The potential energy, which arises from the coulombic attraction between the negative charge of the electron and the positive charge in the nucleus, is given by U Ze2/r. The model we will describe here, due to Niels Bohr in 1913, is an early attempt to predict the allowed energies for single-electron atoms. Furthermore, a related analogy between electrons and planets played a significant role in Bohr’s subsequent articulation of the model. Accessibility Statement For more information contact us at. The elaboration of the negative analogy led to the conclusion that the electron could move only in certain discrete orbits and its energy and angular momentum were accordingly restricted. We will argue that it was the negative, rather than the positive, analogy between intra-atomic electrons and the rings of Saturn that motivated Bohr to adopt and develop Rutherford’s atomic model. And before moving on to the next topics, here are some more practice examples of the Bohr model of the hydrogen atom that you can work on. 
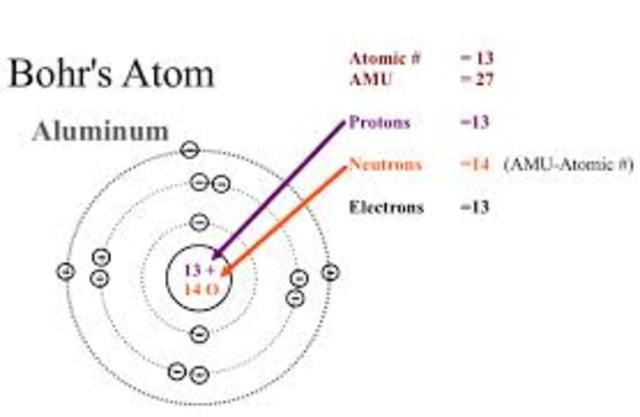
In our paper we will stress the significance of negative analogies for the development of Bohr’s atom. It then went on to derive the energy levels of the electron in a hydrogen atom using the laws of uniform circular. However, she underappreciated the role of negative analogies in model-building. The Bohr model was derived via the classical assumption that the electron orbits the nucleus, much like how the Earth orbits the Sun, except that the origin of the centripetal force was electromagnetic instead of gravitational. Bohrs model suggests each atom has a set of unchangeable energy levels and electrons in the electron cloud of that atom must be in one of those energy levels. The model can be applied to hydrogen or hydrogen-like atoms to explain their line emission spectrum. These orbits have quantized energy and angular momentum associated with electron. Modern scholarship on analogy takes its departure from the work of Mary Hesse, who pointed out the existence of negative analogies between two different physical systems, that is, those respects in which the two systems clearly differ. 10.4: The Bohr Model is shared under a not declared license and was authored, remixed, and/or curated by LibreTexts. Bohr model of an atom states that only those orbits are allowed where angular momentum of electron are integral multiple of n h / 2. Electrons revolve around the nucleus in circular paths. The nucleus carries all the positive charge and most of the mass of the atom. According to him: An atom consists of a very small central core called the nucleus. The significant role of models and analogies in scientific practice has been widely recognized. In (1913,) Neil Bohr explained the stability of the atom based on the observations of his experiments.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
